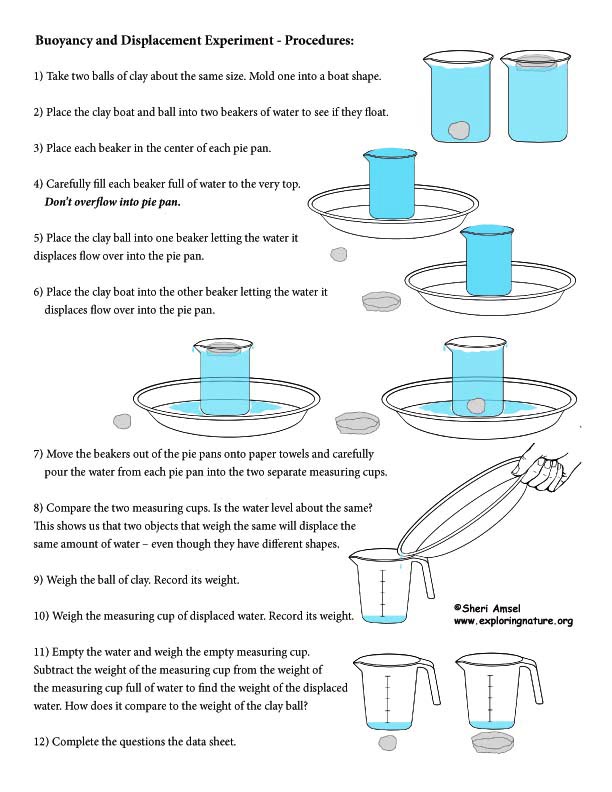
Focus Questions:
1. What is the difference between mass, volume and density?
2. How do you find the volume of a substance?
3. How do you find the mass of an object?
4. How do you find the density of an object?
5. How does the density of an object affect its ability to float?
Volume is how much space an object (or amount of matter) takes up.
Mass is a little more complicated. Mass can be describes as the weight of an object on Earth (where gravity is the same everywhere on the planet). Mass is actually more specific than weight. Mass is really the “quantity of matter” that makes up an object (so it is the same on Earth or on the moon or anywhere in the Universe), but it would weigh less on the moon than on Earth because the moon has less gravity. So, again, mass is really the “quantity of matter” that makes up an object no matter what the gravity.
Density = Mass/Volume, which means that density is how heavy an object is compared to its size – so how closely its molecules are “packed” together.
For instance, generally a rubber ball is less dense than a baseball of the same size. Let’s look at what that looks like below. Each ball has the same volume. The baseball, however, is made up of more “matter” so has more mass per unit of volume. The baseball is denser than the rubber ball – it has more mass per unit of volume than the rubber ball. It has a higher density.
Density = Mass/Volume also means that the larger the volume of an object compared to its mass, the less dense it is.
For example, if a marble weighs the same as a much larger foam ball, the marble has a higher density than the foam ball, despite its smaller size. Let’s look at what that looks like below.
Density and Water
Scientists often equate the density of an object by how it compares to the density of water. Here are some important facts about density and water:
1) When an object is put in a body of water, it displaces its volume (size) of water.
2) If an object floats, it is less dense than water.
3) If the object sinks, it is more dense than water.
That is why a life raft floats (it is less dense than water) and an anchor sinks (it is more dense than water).
Finding the Density of Water Activity
Do this simple activity to find the density of water. Keep this equation in mind: Density = Mass/Volume
Materials:
scale, beaker, water
Procedure:
1) Weigh an empty beaker to find out its mass. (In this example, it will be about 20 gm.)
2) Fill the beaker with 100ml of water. (100ml is the water’s volume.)
3) Then reweigh the beaker with the water in it. (It will be about 120 gm.)
4) Subtract the mass of the empty beaker from the mass of the full beaker and you have the mass of the water.
120 gm – 20 gm = 100 gm.
So you now have the mass of the water and the volume of the water. To find the density of water use the equation:
Density = Mass/Volume
Density = 100gm/100 ml – So water has the density of 1 gram per milliliter (1gm/ml).
Things with a density of less than 1gm/ml will float and those with a density of more than 1gm/ml will sink.
Density Saving Your Life...
We are made mostly of water, so our density is close to that of water, which means that we do float, but so close to the water level that usually not enough of the head will be out to breathe. That is why life jackets are required for boaters.
How does wearing a life jacket allow us to float? Life jackets are made to have very low density, with material that traps air inside. This allows the life jacket, and the people in them, to float.
When you research information you must cite the reference. Citing for websites is different from citing from books, magazines and periodicals. The style of citing shown here is from the MLA Style Citations (Modern Language Association).
When citing a WEBSITE the general format is as follows.
Author Last Name, First Name(s). "Title: Subtitle of Part of Web Page, if appropriate." Title: Subtitle: Section of Page if appropriate. Sponsoring/Publishing Agency, If Given. Additional significant descriptive information. Date of Electronic Publication or other Date, such as Last Updated. Day Month Year of access < URL >.
Amsel, Sheri. "Volume, Mass and Density" Exploring Nature Educational Resource ©2005-2024. December 13, 2024
< http://exploringnature.org/db/view/1825 >