Focus Questions and Goals
1) What is matter?
2) Name the three phases (states) of matter?
3) Identify and describe the particle in each phase of matter and how they are different in each phase of matter.
About Matter
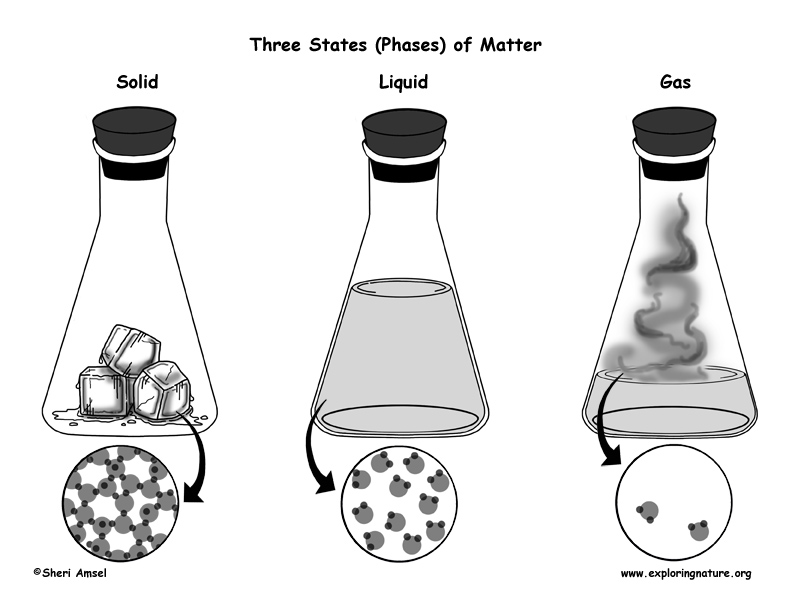
Matter is something that has mass and volume (takes up space). Matter can be found in several phases or states. The three most common phases of matter on Earth are solids, liquids and gases. Less commonly, we can also find matter as plasma or Bose-Einstein (BE) condensate.
Solids have a fixed shape and volume. A solid’s particles are packed closely together. There is not much space between the particles and there is little particle movement. A solid is not easily compressed.
Liquids have a fixed volume, but take the shape of the container in which they sit. There is not much space between the particles, but they can slide past each other and flow easily. A liquid is not easily compressed.
Gas fills the shape and volume of the container in which it sits. There is a lot of free space between its particles and they flow easily past each other. Gas can be compressed.
These are physical states of the molecules of matter. Molecules can shift from one physical state to another without changing their molecular structure (or chemical state). Water is still H20 when it is ice, steam or a liquid – even though its physical state has changed.
Physical states can be changed by adding energy (i.e. increasing temperature or pressure) or releasing energy (i.e. cooling or lowering pressure). This does not change the matter’s molecular structure. It is still the same matter or substance. When you heat a substance, you are adding energy to it. The movement of its molecules will increase until its physical state changes.
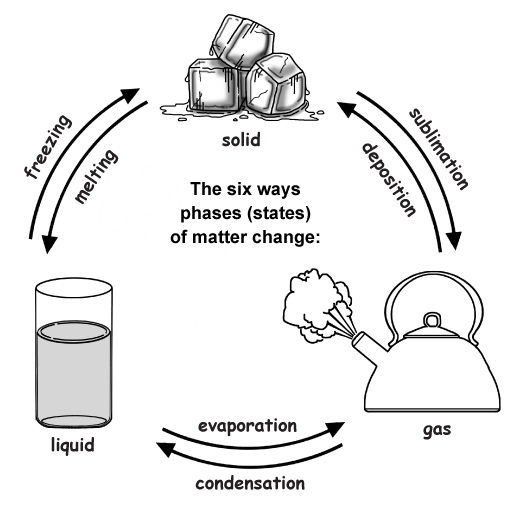
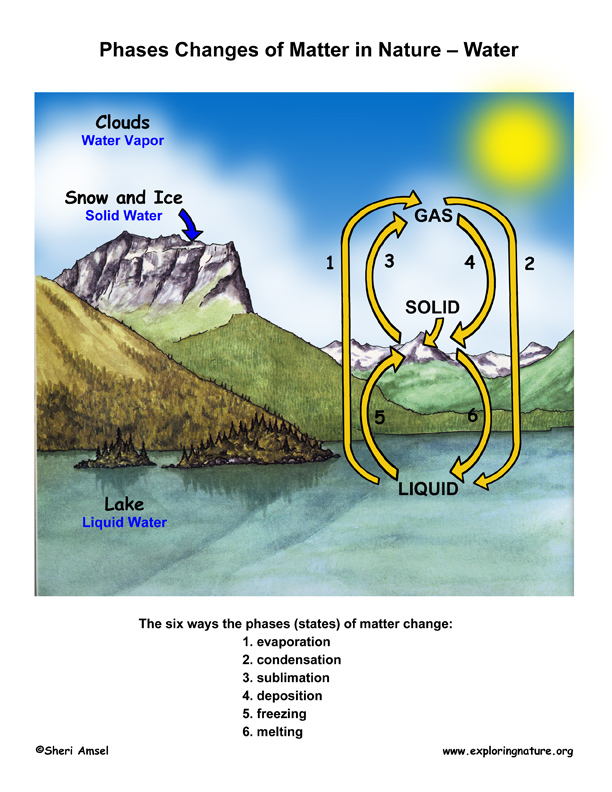
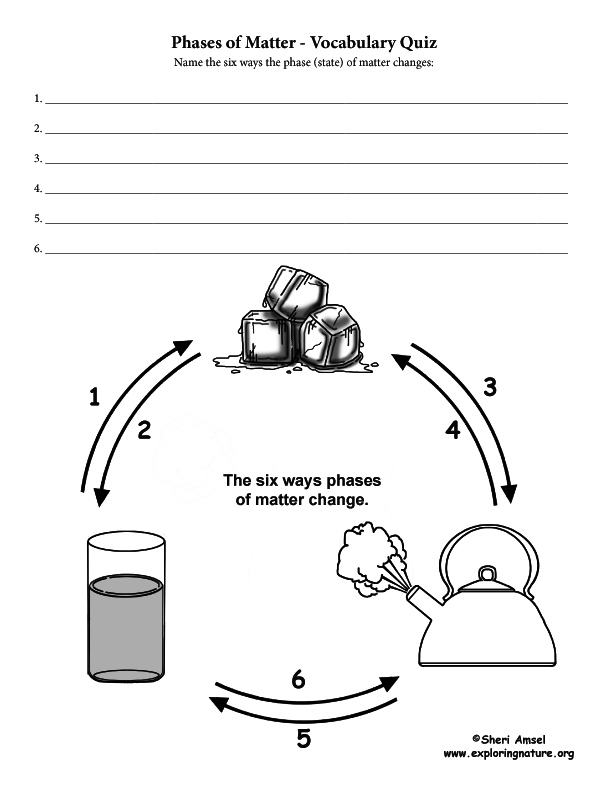
The six ways to change the phase (state) of matter:
1) Melting changes a solid to a liquid. (i.e. dripping icicles)
2) Freezing changes a liquid to a solid. (i.e. lake freezing over)
3) Evaporation changes a liquid to a gas. (i.e. clothes drying on a clothesline)
4) Condensation changes a gas to liquid. (i.e. water forming on the outside of a cold glass)
5) Sublimation changes a solid to a gas. (i.e. ice cubes shrinking in the freezer)
6) Deposition changes a gas to a solid. (i.e. frost forming on the windows)
These changes happen at precise temperatures for different substances. Scientists refer to these as melting point, freezing point, condensing point and boiling point. Water’s melting point is 0°C, while its boiling point is 100°C.
Water’s Unique Quality
Usually solids are more dense than liquids because their molecules are closer. Water is an important exception to this rule. When water freezes, it forms a bonded arrangement of molecules that is actually less dense than liquid water, yet takes up more space. This is an important feature of water in that when ice forms on a lake or other waterway, it is less dense than the liquid water – so forms on top of the liquid water. This allows water animals to stay alive all winter under the ice. Without this trait, many of the living things on Earth would not have survived and evolved to what we have today.
It is also important to note (and helpful to us) that other substances have much higher melting temperature than water. Imagine if salt or sugar melted at room temperature the way water does.
Assessment for Vocabulary and Comprehension Below (2nd PDF):
Link to Phases of Matter Multiple Choice
Link to Phases of Matter Vocabulary Quiz l
Link to Phases of Matter Matching Vocabulary Quiz ll
More Physical Science Activities: LINK
When you research information you must cite the reference. Citing for websites is different from citing from books, magazines and periodicals. The style of citing shown here is from the MLA Style Citations (Modern Language Association).
When citing a WEBSITE the general format is as follows.
Author Last Name, First Name(s). "Title: Subtitle of Part of Web Page, if appropriate." Title: Subtitle: Section of Page if appropriate. Sponsoring/Publishing Agency, If Given. Additional significant descriptive information. Date of Electronic Publication or other Date, such as Last Updated. Day Month Year of access < URL >.
Amsel, Sheri. "Phases of Matter – Gas, Liquids, Solids" Exploring Nature Educational Resource ©2005-2024. December 14, 2024
< http://www.exploringnature.org/db/view/1824 >